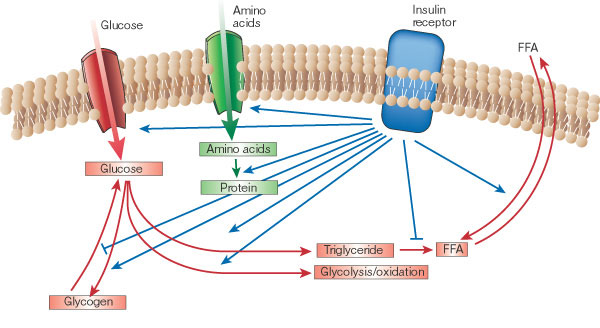
Crack open any physiology textbook and chances are you’ll learn that after eating any normal meal, the release of insulin from the pancreas then signals the shutdown of the release of fatty acids from adipose (body fat) tissue and the increase of fatty acid uptake.
It’s an easy conclusion to make. The logic goes that carbohydrates through their stimulation of insulin are fattening beyond their contribution of energy as kilocalories. It doesn’t matter how much you eat, so long as you avoid carbs to lose weight.
Another growing belief floating mainly around fitness circles is that it’s best to forego foods containing carbs when heading to the gym. It’s for fear that the carbs’ action on insulin will squash fat burning stimulated by exercise. Then again, some low-carb proponents have also argued, physical activity as a means to expend energy for weight management is pointless altogether. Again, carbs are really all that matter because of their action on insulin.
Where does all the extra energy from excess protein and fat go when overconsumed? And what about protein’s own effects in stimulating insulin or insulin’s role in promoting satiety? These questions are often overlooked or not easily answered by those that promote the “insulin is a fat storage hormone” proposition.
Out to help repair insulin’s reputation is obesity researcher Stephan Guyenet, Ph.D., of the University of Washington, who studies regulation of body fat by the brain. He downplays insulin as a primary regulator of long-term fat storage calling it a misrepresentation and oversimplification of what the hormone’s role really is. “There has been a lot of confusion about the role of insulin in the regulation of fat tissue,” Guyenet comments.
“Insulin is a critical coordinator of dynamic fatty acid flux on a meal-to-meal basis,” Guyenet clarifies. Through coordination, insulin is the main hormone that, essentially, tells the body what to do with the food energy just eaten. For example, when carbohydrate or protein is eaten, insulin directs the body to burn the carbohydrate or protein instead of using fat. And when mostly fat is eaten, the lack of an insulin response directs the body to burn the fat just eaten.
“Either way, you burn what you eat, and when that runs out, you go back to burning stored fat,” according to Guyenet. “This process is easily misinterpreted because one of insulin’s main functions following a meal is to shut down fat release from fat cells while the body burns carbohydrate and protein.
From Guyenet’s explanation, it sounds more as if insulin’s role is really that of a traffic cop— signaling where nutrients should go (carbs, protein, and fat) just after meals. Overall, however, insulin does not promote fat accumulation in the long run. Keep in mind that insulin also promotes the transport of glucose and amino acid into muscle for synthesis of glycogen and proteins after meals.
“At the end of the day, the total 24-hour ‘flux’ of fat in and out of fat cells does not appear to depend on these insulin spikes,” Guyenet comments.
Making friends with insulin through exercise
Exercise researcher John Ivy, Ph.D., of the University of Texas, is in agreement that insulin is an often-misunderstood hormone and also makes the point that insulin’s specific action following meals does not relate to long-term fat storage. “The most important thing is calories burned in a day,” he comments.
As previously shared on this blog, Ivy emphasizes that understanding how to make the best use of insulin and carbohydrate can be critical for long-term weight management. The main reason is because of their support to muscle maintenance and growth. Ivy reminds that insulin’s role extends well beyond its action on promoting fatty acid synthesis. The hormone also both prevents protein breakdown and boosts protein synthesis in muscle. Although protein by itself promotes insulin’s release (again, often overlooked by low-carb proponents), carbohydrates can boost both these effects.
Additionally, when carbohydrates are consumed during or after exercise, they can help fuel a better workout overall (as a fitness-model-nutrition-scientist colleague of mine once put it, “working out without eating some carbs makes you feel like @*$”). Carbohydrates also replenish glycogen for faster recovery and better workouts later. Finally, carbohydrates also help reduce levels of the exercise stress-induced adrenal hormone cortisol, which is implicated in protein catabolism, aiding muscle recovery.
The main consideration, Ivy explains, is that “one does not want to lose muscle.” Skeletal muscle can be a primary source of glucose disposal and energy consumption (with exercise) and can play a role in determining metabolic rate. But dieting alone often leads to both fat and muscle loss, which can lead to a reduced metabolic rate and an increasingly harder time continuing to lose weight. Without exercise, the chances of putting weight back on increases and that, in turn, leads to greater risk of insulin resistance. “Dieting alone is not the answer. To prevent muscle loss, one needs to exercise when dieting, which will accelerate fat loss,” he said.
He also emphasized the role of nutrient timing, or consuming nutrients at times that coincide with (e.g. carb/protein drinks) or directly after physical activity, to make the most of insulin. Directly after exercise is when muscles are most insulin sensitive increasing the likelihood that nutrients will be used for muscle protein and glycogen synthesis.
Diabetes researcher Joseph Henson, Ph.D., of University of Leicester, affirms that physical activity’s powerful effects on promoting insulin sensitivity makes it the “first line” of defense for prevention and treatment of insulin resistance. But while perhaps timing nutrients for after exercise may be best, he says that just getting any activity is what counts most. “A single bout of exercise can increase insulin sensitivity for at least 16 hours post exercise in healthy people as well as people with type 2 diabetes,” Henson comments.
On the other hand, the lack of activity, or general muscle contraction, Henson argues, can exacerbate insulin resistance and decrease insulin sensitivity, which are prerequisites for type 2 diabetes. Henson’s recent research suggests that probably more important than exercising regularly is simply avoiding staying sedentary for too long for cardiovascular health.
“We found that it was the amount of time that people spent sitting that had the biggest impact on glucose, triglycerides, and HDL cholesterol and not the level of exercise,” he said. “This just shows what a negative impact too much sitting can have on an individual’s health, independent of the level of exercise.”
With respect to the powerful effects of physical activity on insulin sensitivity and maintenance of skeletal muscle, it’s unclear why “insulin as a fat-storage hormone” proponents would argue against the use of exercise as an additional support for weight management. Of course, one could point out studies that have shown that exercise alone does little for long-term weight loss, but doesn’t that discredit further the idea that insulin is a long-term fat storage regulator?* “This is particularly ironic,” Guyenet explains, “since exercise is probably the most effective way to increase the insulin sensitivity of lean tissue. Exercise can also reduce fasting insulin to some extent.”
Why low carb really works for weight loss
If not because of reduced insulin, how then does a lower-carb diet really work for weight loss? Professor of regulation of food intake Margriet Westerterp-Plantenga, Ph.D., of Maastricht University Medical Centre, suggests that the reason low-carb works has nothing to do with carbohydrates at all, but everything to do with protein and its role on energy balance.
In a review paper last year, Westerterp-Plantega (along with colleagues Sofie Lemmens and Klaas Westerterp) wrote that controlled trials have shown that the answer is that it is the relatively higher protein of the diets including Atkins, South Beach Diet, Paleo, etc., and not the relatively lower carbohydrate content that has led to the success of these approaches for weight loss. The reason is that dietary protein acts on three “metabolic targets”: it increases satiety, stimulates energy expenditure, and spares fat-free muscle (helping to maintain resting energy expenditure).
Westerterp-Plantenga’s hypotheses are now supported further by a new study of which she was the lead author published earlier this year. The study compared two energy-restricted diets, one normal in protein (0.8g/kg/d) and one higher in protein (1.2g/kg/d) on 72 overweight and obese men and women. While both groups lost weight, as expected, the group that consumed more protein retained more muscle and had a higher resting metabolism (this was independent of physical activity).
Along with the study’s findings, which were published in the Journal of Nutrition, Westerterp-Plantenga points out that the protein in both diets facilitated satiety. The study evaluated satiety through measurement of concentration of appetite-regulating hormones glucagon-like peptide 1 (GLP1) and peptide YY 3-36 (PYY).
In addition, another recent study by Anita Belza and her colleagues, of the University of Copenhagen, evaluated different protein intakes on appetite-regulating hormones including GLP-1, PYY, and glucagon. The study was performed on 25 men in a three-way randomized, double blind manner that involved test meals with 30 percent energy from fat and normal protein (14 percent energy), medium-high protein (25 percent energy), and high protein (50 percent energy). The study, published in AJCN earlier this year, found that protein induced satiety in a dose-dependent manner.
These findings might explain why it has been observed in other previous studies, as reported by KJ Acheson, that “for every 1 percent increase in protein intake, replacing fat or carbs, observed decreases of energy intake range from 32 to 51 kilocalories.” That’s some satiety. In addition, he wrote that animal protein stimulates 24-hour energy expenditure slightly but significantly more than vegetable proteins (likely due to animal protein’s content of branched-chain amino acids and action on insulin).
All of this research support Guyenet’s assertions that fat regulation probably has a lot more to do with appetite than with insulin. “Insulin regulates fat cell function in a dynamic, meal-to-meal manner, but it is probably not a major regulator of total fat tissue size (despite its ability to alter body fatness in extreme circumstances like type 1 diabetes and insulin injections into the same spot every day),” he writes.
Leptin
In his final thoughts on what is really involved in controlling long-term fat storage, Guyenet adds, “The primary known regulator of body fatness is the hormone leptin.” How does leptin work to regulate fat storage? So far, what is known is that leptin inhibits food intake by affecting long-term appetite, although it doesn’t appear to have any effects on short-term satiety signals (as protein does).
Leptin acts specifically by signaling the hypothalamus that energy stores are sufficient. Notably, when leptin is absent (as in genetically altered mice and humans with rare mutations) it can lead to severe obesity. The effects of leptin in humans are not as well understood; however, obesity is thought to involve a resistance to the effects of leptin on appetite centers.
For more of Guyenet’s thoughts on carbs and insulin, you can head over to his blog. And, you can expect to hear more from Guyenet on the topic of leptin soon.
In the meantime, it’s safe to say that we should all lay off the naming and blaming of insulin for weight gain and obesity. Instead, it’s probably best to continuing to watch energy intake and expenditure, as well as making sure to include physical activity for improved muscle maintenance and insulin sensitivity, for long-term weight management.
*Note: Ivy suggests that to make the most of physical activity for weight loss, it’s important to get enough each day for its additional effects on appetite in the brain that could help diminish overeating. ”When physical activity declines below a threshold level of about 60 percent of resting metabolic rate, the relationship between caloric expenditure and appetite becomes uncoupled. The further one moves below this threshold level of caloric expenditure, the more one overeats,” he said.
References
Acheson KJ. Diets for body weight control and health: the potential of changing the macronutrient composition. Eur J Clin Nutr. 2012 doi: 10.1038/ejcn.2012.194.
Belza A, et al. Contribution of gastroenteropancreatic appetite hormones to protein-induced satiety. Am J Clin Nutr. 2013 doi: 10.3945 / ajcn.112.047563
Henson et al. Associations of objectively measured sedentary behaviour and physical activity with markers of cardiometabolic health. Diabetologia. doi: 10.1007/s00125-013-2845-9.
Saltiel AR, Kahn CR. Insulin signalling and the regulation of glucose and lipid metabolism. Nature. 2001. doi: 10.1038/414799a
Soenen S, et al. Normal Protein Intake Is Required for Body Weight Loss and Weight Maintenance, and Elevated Protein Intake for Additional Preservation of Resting Energy Expenditure and Fat Free Mass. J Nutr. 2013. doi: 10.3945 / jn.112.167593
Westerterp-Plantenga MS, et al. Dietary protein – its role in satiety, energetics, weight loss and health. Br J Nutr. 2012 Aug;108 Suppl 2:S105-12. doi: 10.1017/S0007114512002589.
Wolfe R. The underappreciated role of muscle in health and disease. Am J Clin Nutr. 2006;84:475-82.